
Adverse Drug Reaction Classification System

Adverse Drug Reaction Classification System
| Drug ID |
Drug Name
|
CAS RN
|
Molecular Formula
|
Structure
|
| BADD_D00091 | Aluminum oxide | 1344-28-1 | Al2O3 |  |
| BADD_D00092 | Aluminum sesquichlorohydrate | 1327-41-9 | AlClH3O2+ | 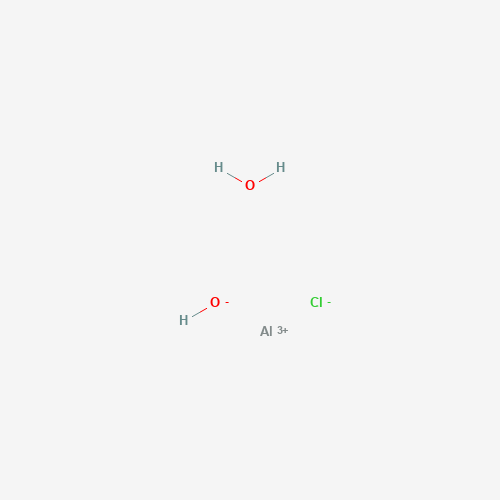 |
| BADD_D00093 | Aluminum tetrachlorohydrex gly | Not Available | Not Available |  |
| BADD_D00094 | Aluminum zirconium | Not Available | Al2Zr |  |
| BADD_D00095 | Aluminum zirconium octocholrohydrex | Not Available | Not Available |  |
| BADD_D00096 | Alvimopan | 156053-89-3 | C25H32N2O4 | 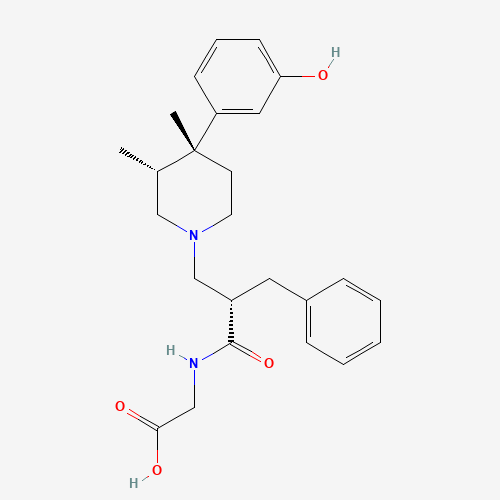 |
| BADD_D00097 | Amantadine | 768-94-5 | C10H17N | 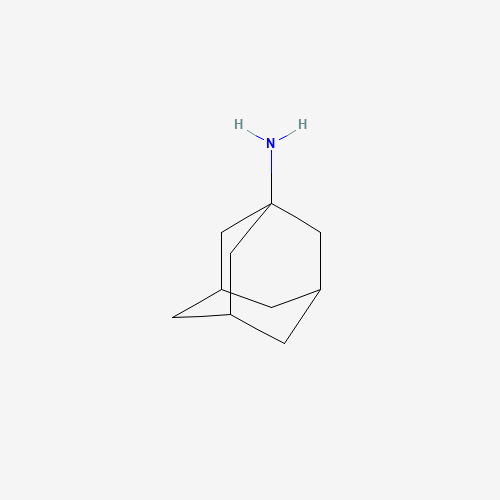 |
| BADD_D00098 | Amantadine hydrochloride | 665-66-7 | C10H18ClN | 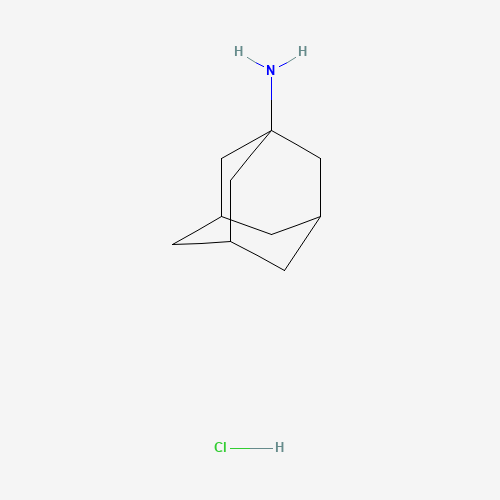 |
| BADD_D00099 | Ambenonium chloride | 115-79-7 | C28H42Cl4N4O2 | 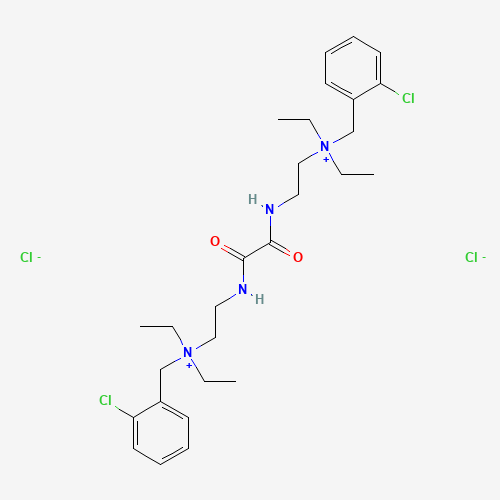 |
| BADD_D00100 | Ambrisentan | 713516-99-5 | C22H22N2O4 | 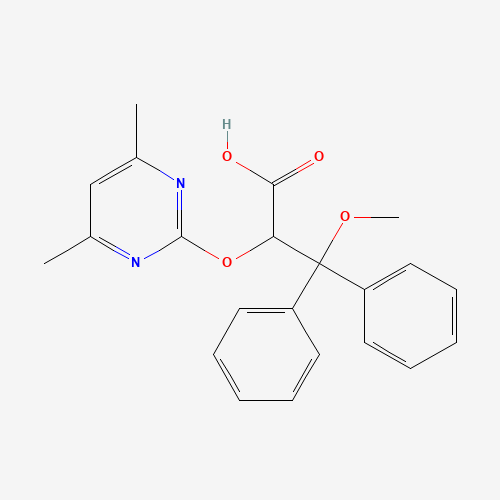 |
| BADD_D00101 | Amcinonide | 51022-69-6 | C28H35FO7 | 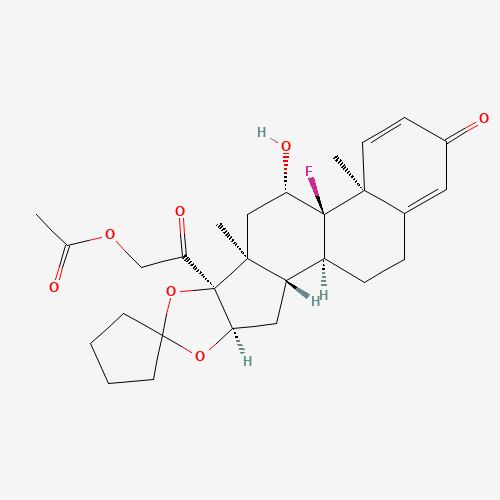 |
| BADD_D00102 | Amifostine | 20537-88-6 | C5H15N2O3PS | 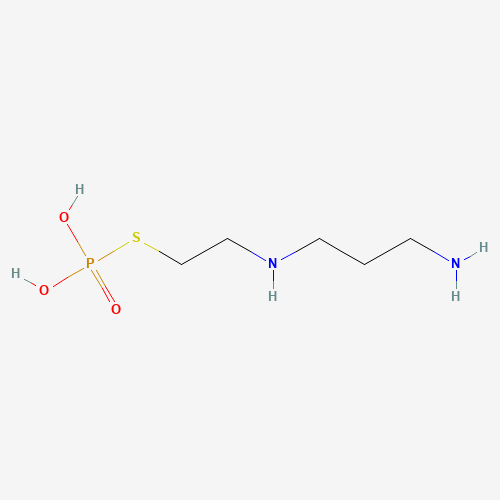 |
| BADD_D00103 | Amikacin | 37517-28-5 | C22H43N5O13 | 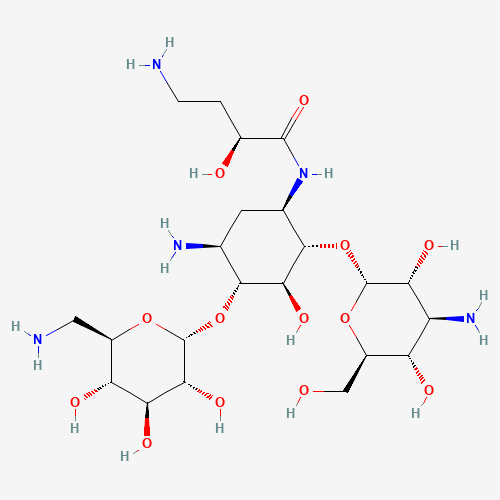 |
| BADD_D00104 | Amikacin sulfate | 39831-55-5 | C22H47N5O21S2 | 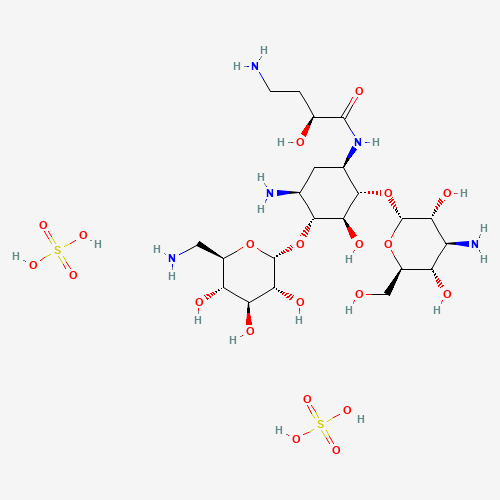 |
| BADD_D00105 | Amiloride hydrochloride | 2016-88-8 | C6H8ClN7O.ClH |  |
| BADD_D00106 | Aminocaproic acid | 60-32-2 | C6H13NO2 | 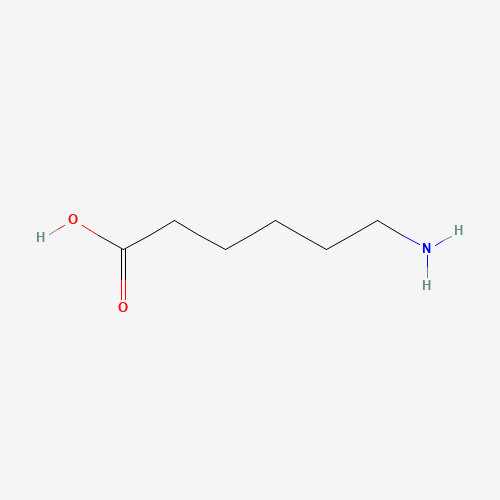 |
| BADD_D00107 | Aminoglutethimide | 125-84-8 | C13H16N2O2 |  |
| BADD_D00108 | Aminohippurate sodium | 94-16-6 | C9H9N2NaO3 | 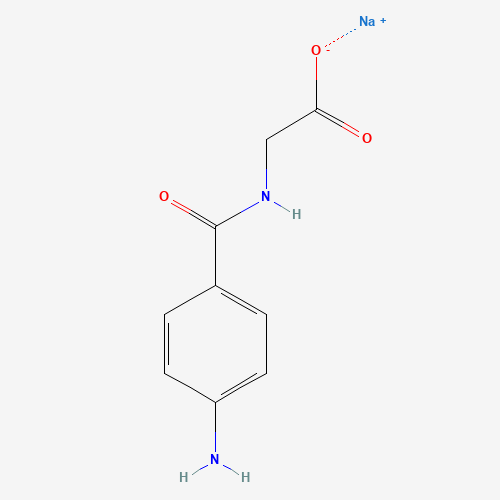 |
| BADD_D00109 | Aminohippuric acid | 61-78-9 | C9H10N2O3 | 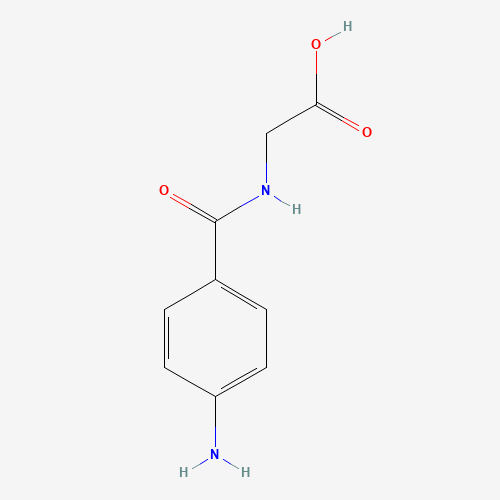 |
| BADD_D00110 | Aminolevulinic acid | 106-60-5 | C5H9NO3 |  |
| BADD_D00111 | Aminolevulinic acid hydrochloride | 5451-09-2 | C5H10ClNO3 | 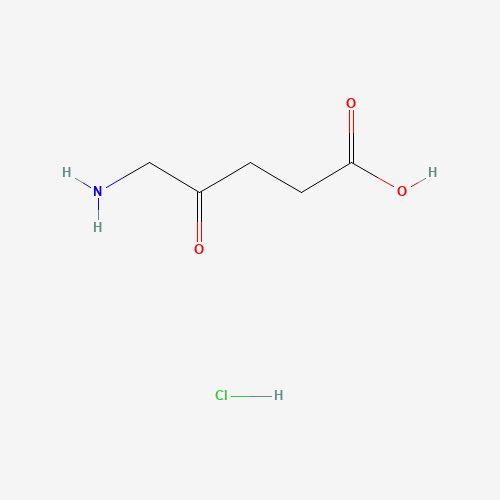 |
| BADD_D00112 | Aminophylline | 317-34-0 | C16H24N10O4 | 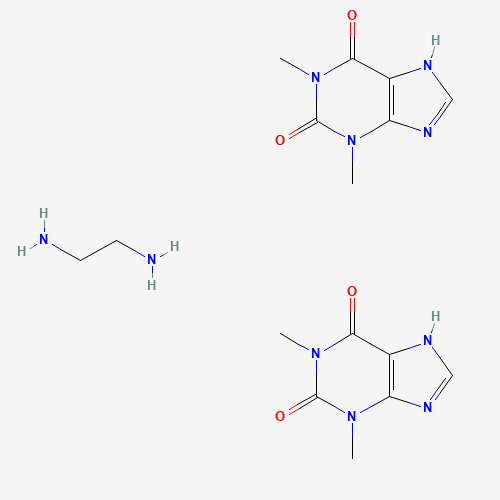 |
| BADD_D00113 | Aminophylline dihydrate | 5897-66-5 | C16H28N10O6 | 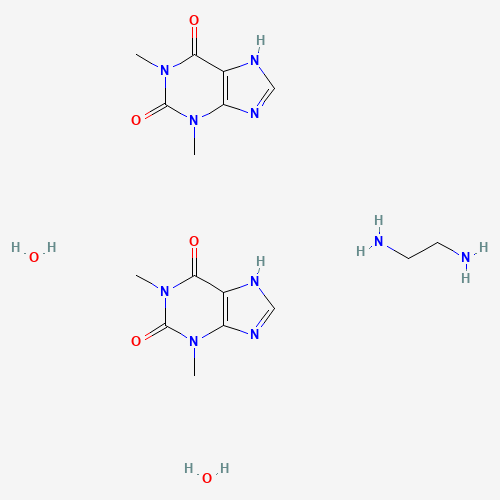 |
| BADD_D00114 | Aminosalicylic acid | 65-49-6 | C7H7NO3 |  |
| BADD_D00115 | Amiodarone | 1951-25-3 | C25H29I2NO3 | 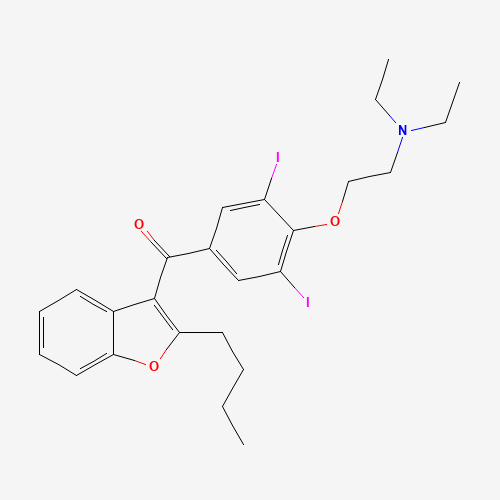 |
| BADD_D00116 | Amiodarone hydrochloride | 19774-82-4 | C25H30ClI2NO3 | 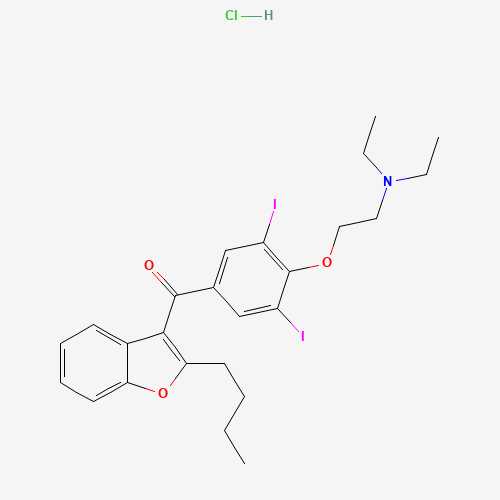 |
| BADD_D00117 | Amisulpride | 71675-85-9 | C17H27N3O4S | 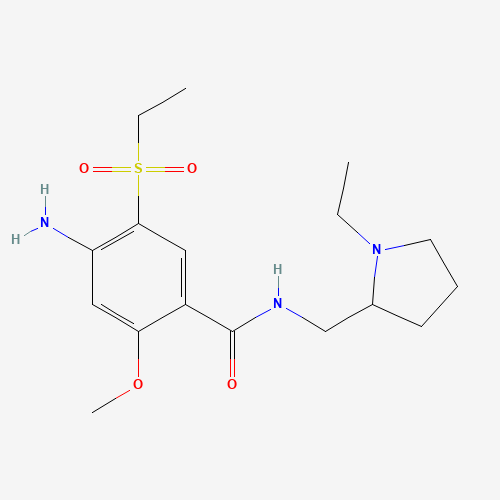 |
| BADD_D00118 | Amitriptyline | 50-48-6 | C20H23N | 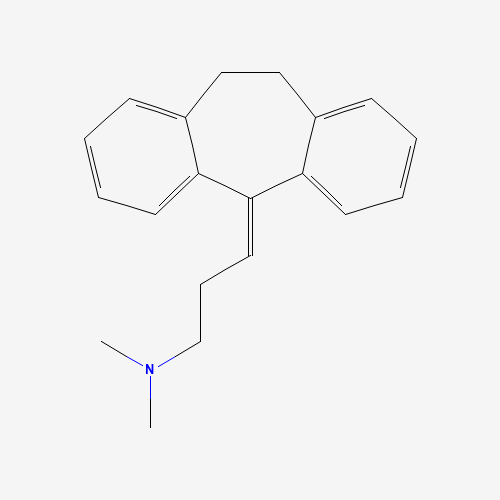 |
| BADD_D00119 | Amitriptyline hydrochloride | 549-18-8 | C20H24ClN |  |
| BADD_D00120 | Amlexanox | 68302-57-8 | C16H14N2O4 |  |